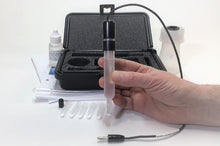
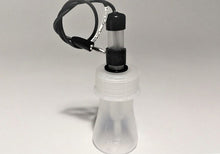
FEATURES:
- Carry Case
- NO LIQUID. The customer is to supply solvent.
- Vial of Silver Nitrate
- Laboratory Electrode Storage Bottle
- Pack of 5 Replacement Porous Tips
- 5 Heat-Shrinkable Sleeves
- Electrode Filling Pipette
- Absorbent Foam
- Maintenance Manual
- One non-choridized silver wire electrode: removable top cap, electrode tube assembly, electro-porous KT glass tip, belch hole with closure
- Vial (1006VG) contains 1 gram Silver Nitrate powder
- Serial number #1006-VG
STEPS TO VERIFY VIABILITY OF ELECTRODE:
- Create a ~5mm layer of electrode solution in a beaker
- Place two electrodes of the same type into the pool of solution
- Compare the readings using a volt meter
- If they are within 10 mV of variation, it is good to go!
HOW IT WORKS:
I need an anhydrous electrolyte, so an aqueous reference electrode is unsuitable. What are the alternatives?
If contamination by water from aqueous electrodes is a problem, there are a number of alternatives. The simplest is to use a salt bridge (Koslow Part No. SB) containing the anhydrous electrolyte to separate the aqueous reference electrode from the analyte solution. Other alternatives include using a non-aqueous reference. The Koslow non-aqueous reference electrode (Part No 1006) requires user assembly and consists of a silver wire immersed in a solution containing silver nitrate dissolved in a solution of an appropriate electrolyte. Ideally, this electrolyte is the same as that used for the analyte (to eliminate junction potentials). An acetonitrile-based electrolyte can generally be used. The potential of the non-aqueous reference electrode depends on the solvent, the electrolyte, and the concentrations of silver nitrate and salt. Since the potential of a non-aqueous reference electrode can vary among different electrodes, redox potentials measured using such a reference electrode should be quoted relative to an internal reference compound (e.g., ferrocene). A pseudo-reference electrode is simply a platinum or silver wire immersed in the analyte solution. This has the advantage that there can be no contamination of the analyte, but the disadvantage is that the reference potential is unknown, as it is dependent on the composition of the analyte solution. Therefore, redox potentials measured using a pseudo-reference electrode should again be quoted relative to an internal reference compound such as ferrocene.
LEARN MORE: