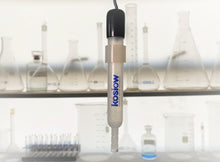
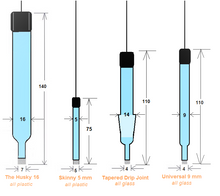
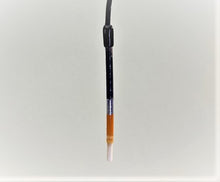
Mercury/ Mercurous Oxide Reference Electrode (Hg/HgO)
Electrode is fully assembled and filled with 4.24 Molar (20% KOH)
- The Hg/HgO is the best choice reference for alkali or fluoride media
- All plastic body parts
- Our standard off-the-shelf concentration of the electrode filling solution is 20% KOH (4.24 Molar) Potassium Hydroxide
- The Mercury Oxide Reference Electrode is ideal for battery development, energy systems research and development, process control, and electro-analytical studies.
- * (NaOH) Sodium Hydroxide electrolyte is also available
- Electrode potential is stable and recovers quickly from small temperature excursions.
- Porous junction tip is made with electroporous plastic- Low noise, low impedance; Prime ion transfer and is replaceable when damaged or contaminated.
Packaged with care in a handy storage carton and includes instructions with SDS. Traceable ID tags on the lead wire.
Electrode Potential:
The standard electrode potential of the Mercury/Mercury Oxide half-cell is +0.098 volts vs. the normal hydrogen electrode (NHE) in basic solution. The standard concentration of the electrode filling solution is 20% (4.24 Molar) Potassium Hydroxide (KOH). The electrode can be ordered in any concentration of Potassium Hydroxide not to exceed 30% KOH due to shipping regulations. Electrode potential is stable to plus or minus one millivolt and recovers quickly from small temperature excursions (about 125° C).
Learn More: